NIST develops on-site detection of fentanyl analogues and opioids to provide reliable compound screening during drug seizures
Source for feature photo: “Drug Seizure” by dvids. The appearance of U.S. Department of Defense (DoD) visual information does not imply or constitute DoD endorsement.
We are in the midst of an opioid crisis. Every day over 130 people die from opioid addiction (National Institute of Drug Abuse) brought on by the misuse of legal drugs like OxyContin®, illicit drugs like heroin, and the drug that straddles the two categories, fentanyl.
Pharmaceutical companies make fentanyl as an alternative pain reliever for cancer patients, as it is 50 to 100x more potent than morphine (Fig 1); however, clandestine labs also make fentanyl and sell it in a mixture with heroin to increase the drug’s potency. This high potency property of fentanyl means that even skin exposure can be life-threatening; roughly 2 milligrams of the compound is lethal, whether by inhalation, ingestion, or skin absorption.
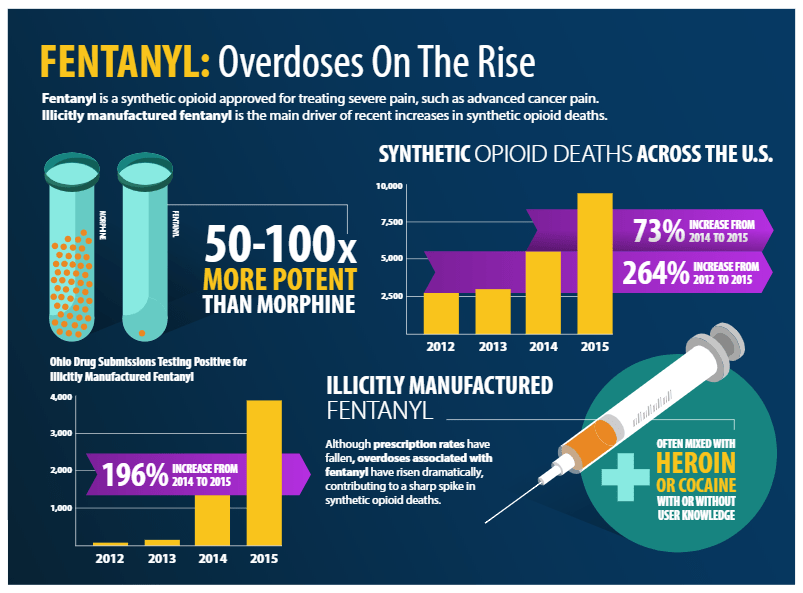
A great concern is that police are exposed to fentanyl. A lot. The DEA published that in just one year, law enforcement in Ohio reported approximately 4000 crimes involving fentanyl drugs or their analogues.
To protect everyone handling these potentially-life threatening drugs, police offices adopted highly vigilant practices (Fig 2); anyone at the scene treats the drugs as non-pharmaceutical fentanyl until confirmed otherwise by laboratory analysis. However, these uber-vigilant practices are undesirable for multiple reasons; namely, that they are cumbersome, expensive, and create copious amounts of stress for those involved.

That is why NIST, the National Institute of Standards and Technology, set out to create a rapid and safe screening test for fentanyl, validated in realistic scenarios like heroin-fentanyl mixtures. Using their technique, officers could perform the field testing before directly handling the sample and determine if these uber-vigilant practices were necessary. Though previous field techniques focused on color tests, which have a variety of disadvantages including requiring large sample sizes and subjective interpretation of color results, the NIST team focused on two instrumental techniques: ion mobility spectrometry (IMS) and thermal desorption direct analysis in real time mass spectrometry (TD-DART-MS).
Transitioning to instrumental rather than chemical techniques presented two challenges. First, NIST needed to determine if these laboratory instruments could be taken to the field. At the time of the study, a field-deployable IMS already existed, and TD-DART-MS showed great promise for the lab-to-field transition. As the CDC advises having a two-tiered testing system, qualitative analysis on IMS followed by quantitative analysis on TD-DART-MS would fulfill the requirement.
Next step; the NIST team, located in the Surface and Trace Chemical Analysis Group, tackled the detection limit (how little compound can be provided for a positive identification) and compound selectivity (only positively reacting to the compound of interest) of the two systems to detect trace amounts of fentanyl located on the exterior packaging of seized drugs.
To that end, the authors Sisco et al. evaluated 17 fentanyl and fentanyl analogues (drugs that have similar effects to fentanyl but have slightly different chemical structures) along with five other opioids (e.g. heroin, methadone, naloxone) and filler substances found in street heroin. Notably, Sisco et al. used a wipe as the collection substrate, showing how in the field, first responders could take a simple wipe to the outside of a drug container and perform the screening procedure. Upon testing their calibrated systems, they found that the IMS and TD-DART-MS systems could positively identify fentanyl at even picogram (1000x less than nanogram or 100000x smaller than a spec of dust in your hand) quantities.
Furthermore, when evaluating mixtures, Sisco et al. wondered if all these compounds mixed together would inhibit detection of fentanyl by interfering with the molecule’s ionization. Both IMS and TD-DART-MS first energize the sample to create ions i.e. the electrically charged versions of molecules. However with complex mixtures, many molecules compete for the limited energy introduced to the sample. If too many other molecules become energized, then their signals can overlap or overpower the fentanyl signal. Therefore, determining how specific compounds are affected by competitive ionization was key to ascertain if IMS and TD-DART-MS can identify fentanyl and its chemical analogues in street drugs.
Sisco et al. evaluated different compound mixtures with fentanyl with IMS and TD-DART-MS. For TD-DART-MS, acetaminophen, caffeine, quinine (mixed with heroin to form a street drug called Scramble, Fig 3), novocaine, and heroin all suppressed the fentanyl signal. Fortunately, even at a 1:1000 ratio (with one part fentanyl to one thousand parts “other”), the fentanyl signal could not be entirely obscured. Other fentanyl analogues demonstrated similar trends to fentanyl, and the NIST team concluded competitive ionization is not an issue for fentanyl identification by TD-DART-MS.

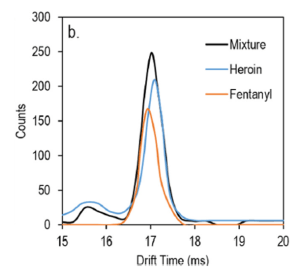
For IMS, Sisco et al. found that most of their competing compounds did not hinder fentanyl signal generation, but novocaine completely eliminated the fentanyl signal when present in quantities slightly more than fentanyl. Novocaine, besides a known abused substance, is almost structurally identical to cocaine, and therefore these drugs could cause the same ionization inhibitory effect. Furthermore, the signal peak for fentanyl and heroin on IMS almost completely overlapped, creating a potentially large problem; a working technician could falsely identify fentanyl when heroin is present. Sisco et al. therefore deconvoluted, or separated, the fentanyl and heroin signals in a way to distinguish them (Fig 4), much in the way Photoshop can improve resolution by de-pixelating a digital photograph. This was additionally necessary as four out of 16 tested fentanyl analogues displayed the same effect. Using this deconvolution technique, scientists positively identified fentanyl (and not heroin) in the remaining samples.
The NIST team demonstrated two, back-to-back detection methods for fentanyl that could help police qualitatively identify when fentanyl is present with high confidence. Though emergency personnel and law enforcement across the country will not be rolling out these measures immediately (due to budget constraints, training requirements and more), first responders and their loved ones can rest assured that federal researchers are developing the means to protect and defend those who protect and defend us every day.
| Title | Rapid detection of fentanyl, fentanyl analogues, and opioids for on-site or laboratory-based drug seizure screening using thermal desorption DART-MS and ion mobility spectrometry |
| Authors | Edward Sisco, Jennifer Verkouteren, Jessica Staymates, Jeffrey Lawrence |
| Journal | Forensic Chemistry |
| Publisher | Elsevier |
| Year | 2017 |
| Link | https://www.sciencedirect.com/science/article/pii/S2468170917300152 |
