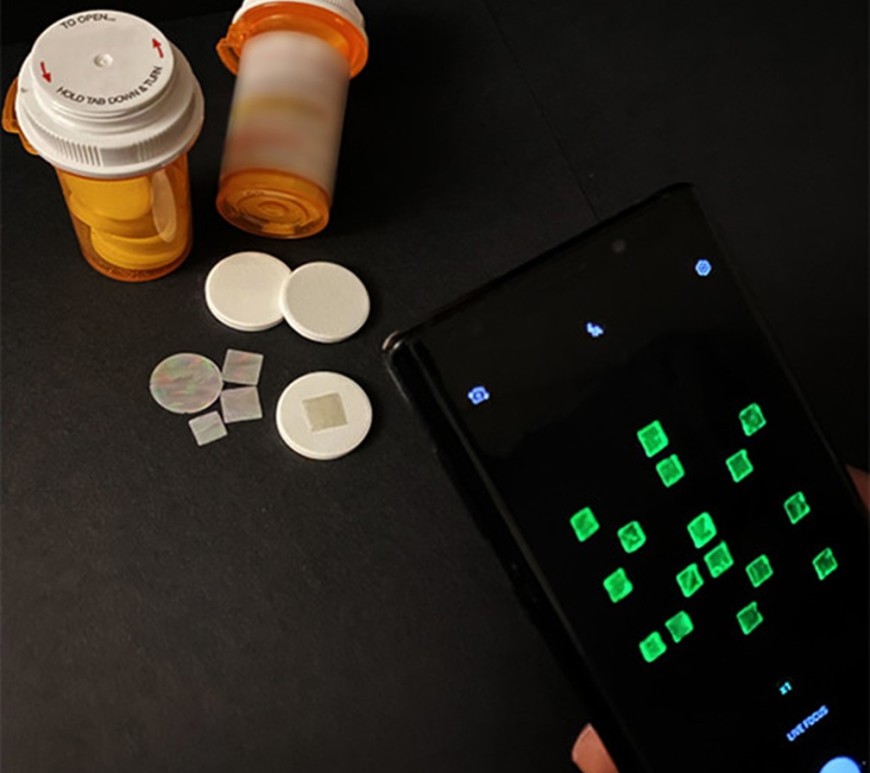
Developing a New Visual Model for Glass Particle Transfer
Glass particles are notoriously difficult to study since they are small and nearly invisible. A Dutch research group is using glow-in-the-dark sand to study glass particle transfer. Continue reading Developing a New Visual Model for Glass Particle Transfer