“Untitled” by Jhayne is licensed under CC BY-NC-SA 2.0
Cocaine is a highly addictive stimulant made from the leaves of the coca plant. Often found as a white powder, this drug poses a serious health risk to first-time users and addicts alike.
Forensic scientists detect cocaine in evidence using two methods. First, if a white powder is found by police, police test the substance using pre-packaged chemicals that change color when they come into contact with certain drugs (Fig 1). Most concerning, these “color tests” have high rates of false positives. After performing the initial, presumptive test, the unknown sample is then sent to a forensics laboratory for confirmatory analysis. Second, if an officer suspects someone of being under the influence of a substance other than alcohol, they will bring that person in for toxicological analysis, where a laboratory technician will collect blood or urine samples to send for confirmatory analysis.
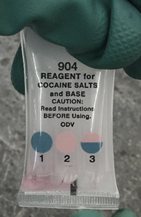
There are two major problems with these methods. First, the confirmatory analysis is destructive and prevents further downstream tests. Second, the samples undergo extensive, laborious and expensive sample pre-treatment contributing to evidence backlog.
To address these problems, researchers began to look at identifying drug usage through metabolites. As a drug moves through the human body, the original compound interacts with various biological systems and changes into different forms. These changed forms of the drugs are called metabolites. In saliva, cocaine does not go through the process because the enzymes needed to metabolize cocaine, usually found in the liver, are not present in your mouth. This means that cocaine only exists in one form for forensic investigators to find. Additionally, technicians collect saliva with mouth swabs, which is significantly less invasive for the donor and less awkward, then a urine or blood sample.
To solve these two problems, researchers in Dr. Igor Lednev’s lab at SUNY Albany in partnership with the University Institute of Research in Police Sciences at the University of Alcala investigated the application of ultraviolet resonance Raman spectroscopy (RRS) to detect cocaine in saliva. This technique collects the unique vibrations generated by chemical compounds when exposed to UV light (wavelengths of 400-10 nm) at a wavelength specific to the compound of interest.
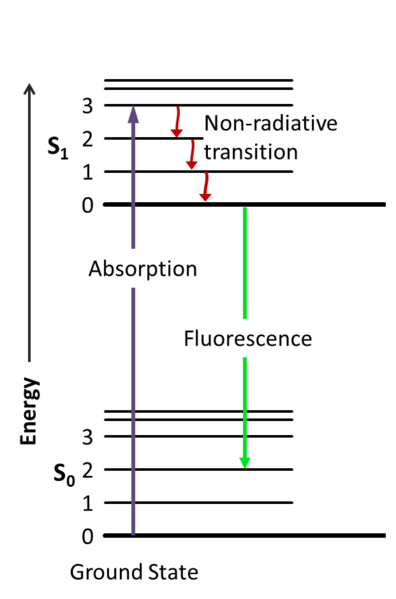
Until recently, Raman spectroscopy was not used in forensics because of its difficulty detecting very small amounts of substances, like trace evidence. However, to augment the low sensitivity plaguing the introduction of Raman spectroscopy, resonance Raman spectroscopy targets wavelengths close to the electronic transition of a compound. Electronic transitions are when light interacts with matter and temporarily gives the electrons more energy or exciting them into an electronic transition to a higher energy (Fig 2). The wavelength that this happens at is specific to the compound, but not unique. To further break this down; electronic transitions will only happen around a certain wavelength for a compound, however, there are many compounds in existence that also absorb energy at the same wavelength. In comparison, normal Raman spectroscopy usually scans compounds with a standard 785 nm wavelength.
However, scientists must validate RRS’ detection limit before introducing the method into the forensics field. Most analytical scientists know that a method is only as good as the detection limit in a real-world sample matrix. The method’s detection limit (LOD) describes the smallest amount of a substance that has a detectable signal over the background noise. Noise is any “signal” that is not from the substance being measured, predominantly created by the background substrate (such as the glass slide the cocaine sample is dried and analyzed on). Often, forensic scientists analyze chemical evidence in trace amounts, and the SAMHSA, or the Substance Abuse and Mental Health Services Administration, defines the cut-off values acceptable for a method’s LOD. The government publishes an updated list of cut-off values every year, where each level is specific to the test used, the compound of interest, and the sample matrix, oftentimes a body fluid like blood. The cut-off level for cocaine in oral fluid is 8 ng for 1 mL of saliva. This amount is roughly equivalent to 8 pinches of salt in 10 tons of potato chips. Every new method submitted to nation-wide use must meet or exceed these cut-off levels to be considered effective. The job of the study’s lead scientist, PhD candidate Valentina D’Elia, was to determine the detection limit for her new method.
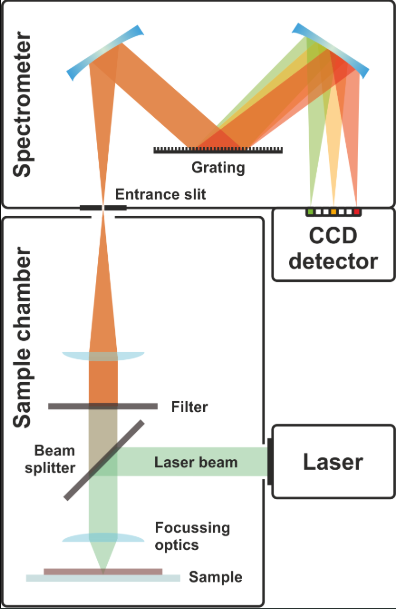
D’Elia first measured the signal given by pure saliva using a laser set to the 239 nm UV wavelength. Most things in the world give off a Raman signal, even us! This creates a large background or noise response that can obscure signals from the cocaine. Therefore, D’Elia did not directly monitor the cocaine-specific signals. Instead, she created a difference spectrum, subtracting the cocaine-specific Raman peaks from the Raman signals from saliva. This allowed D’Elia to eliminate significant background interference and pinpoint peaks relevant only to cocaine and determine the method’s detection limit.
Determining the detection limit is more technical than simply finding the lowest concentration that produces a signal. The signal-to-noise ratio (S/N) quantifies how well the signal of the compound of interest can be distinguished from the background noise that is always present. For the detection limit, the S/N must be around three. D’Elia determined the LOD for cocaine in saliva UV Resonance Raman at 239 nm to be approximately 10 µg/mL. The spectra for the full range of concentrations is shown (Fig 4) to demonstrate how the cocaine-specific signals become more difficult to distinguish from the noise as the concentration of cocaine decreases. At 50 μg the cocaine signals have the largest S/N ratio, whereas at 1 μg there are no distinguishable peaks and only noise.
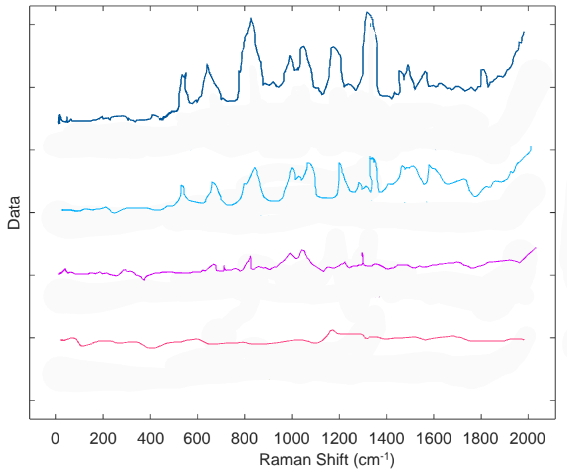
The LOD is significantly larger (almost 4 orders of magnitude) than the cut-off value of 8 ng in 1 mL, and therefore the method is not yet ready for forensic purposes. However, this is not the end of the story for UV resonance Raman spectroscopy. A LOD of 10 μg is a vast improvement over previous techniques, and there are new methods for enhancing Raman signal that researchers around the world are already looking into. Optimizing this and other Raman spec-based methods will provide forensic scientists with analytical techniques with the traditional sensitivity of current forensic methods and the non-destructive, specific, simplicity of Raman.
| Title | Ultraviolet resonance Raman spectroscopy for the detection of cocaine in oral fluid |
| Authors | Valentina D’Elia, Gemma Montalvo, Carmen García Ruiz, Vladimir V. Ermolenkov, Yasmine Ahmed, Igor K. Lednev |
| Journal | Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy |
| Publisher | Elsevier |
| Year | 2017 |
| Link | https://www.sciencedirect.com/science/article/abs/pii/S138614251730567X |

One thought on “The Case for the Detection of Cocaine in Saliva”