PhD student Francesco Sessa and his colleagues at the University of Foggia, Italy have determined that adhesive tape is the best method for retrieving touch DNA left on clothing- even after only 2 seconds of contact.
Feature Image: “fashion, belt, woman, hands, dress, clothes, shopping, retail, clothing, shop” from Pxfuel.
Touch DNA has gained a lot of interest in the criminal justice field and has been used in such notable cases as the deaths of JonBenet Ramsey and Caylee Anthony. Touch DNA comes from skin cells a person sheds when they interact with another person or object. This happened very easily, so touch DNA can be highly prevalent at a crime scene, and thusly very valuable. However, the microscopic nature of skin cells makes them easy to miss and hard to collect. As such, it is very important for scientists to know how to properly collect this evidence to obtain a full DNA profile. Francesco Sessa and his colleagues sought to compare three common methods for DNA collection to identify best method for collecting touch DNA. They determined that the best DNA profiles were obtained using adhesive tape, and that the time of contact between a perpetrator and clothing does not have a significant effect on collection.

While there has been extensive research on the basic principles of touch DNA, there have only been few discussing touch DNA left on clothing. Three main methods employed to collect this kind of evidence are “swabbing,” “cutting out,” and adhesive tape (Figure 2). In the “swabbing” technique, a sterile dry or wet cotton swab is used to dab at the fabric to lift evidence. In the “cutting out” technique, a small subsection of the fabric that is believed to contain evidence is removed. Lastly, the adhesive tape technique calls for attaching a piece of adhesive tape to the fabric and lifting the evidence off with the tape.

Because of the importance of collection method on trace evidence, Sessa and his colleagues have compared these three methods in how they affect DNA profiling. The researchers also investigated the effects of “handling time,” or how long a person is in contact with an object. Ten female volunteers (labeled “wearers”) were given sterilized brassieres to wear for over 12 hours. Then, ten male volunteers (labeled “handlers”) were given the clothing and were instructed to hold it at various locations for times ranging from 2 to 60 seconds.
Sessa et. al. performed standard DNA profiling methodology, by first purifying the DNA, then quantifying, amplifying, and finally, testing the DNA with capillary electrophoresis.
The researchers detected male handler DNA as the dominant or only contributor in almost all 240 samples. Only when adhesive tape was used did the DNA profiles show contribution from the female wearer. Notably, in 5 samples collected using adhesive tape, the wearer contributed the most to the profile. The researchers observed that the wearer profiles were more pronounced when the amount of DNA extracted was low, about 70 pg or below. At DNA concentrations above this, the handler profile was stronger. This indicated to them that a handler’s DNA can “overwrite” the DNA of the wearer when it has a high enough concentration.
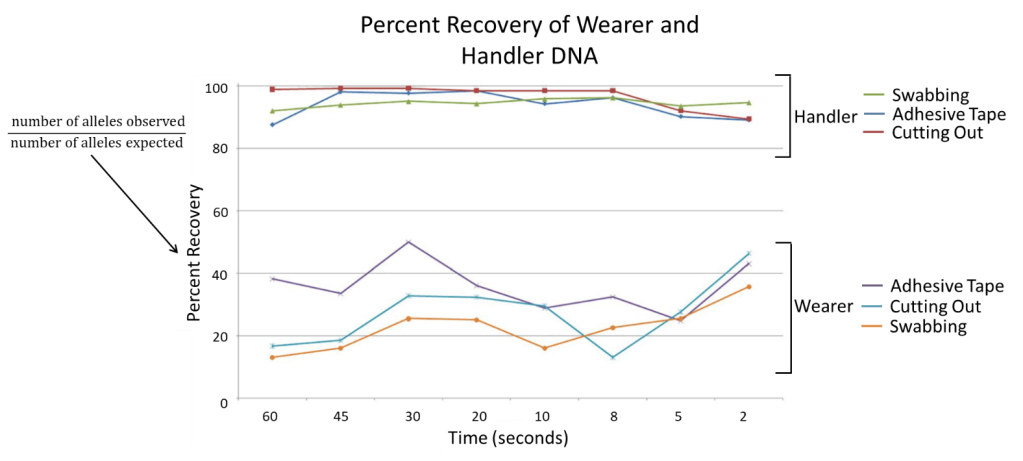
The researchers also determine the actual DNA recovery compared to their expected, called percent recovery, by comparing the total alleles observes for each sample DNA profile to their reference profiles. The team recovered over 87% of the handler DNA with all three methods, but the recovery percentage decreased with shorter times (Figure 3). This demonstrated that even 2 seconds of contact with clothing can leave enough DNA for scientists to obtain a person’s full profile.
For the wearer, percent recovery remained below 50% for all collection methods, with the greatest recoveries obtained from the adhesive tape method. This is useful to know in cases of assault or murder where the perpetrator left no other sources of DNA like blood or semen behind and the technicians must determine whose DNA it is more important to collect: the wearer’s or the handler’s.
Sessa and his colleagues also noted the discrepancy with their work and that of casework and other research, such as those conducted by Poetsch et. al. and Hess & Haas. Technicians often found the wearer DNA to be more prevalent in clothing. In these cases, a much larger piece of clothing was usually tested (about 3-4 cm2 vs. the 1.5 cm2 used in this study), given that the investigators did not know where exactly the clothing was handled. This discrepancy in DNA collected from small and large pieces of clothing illustrates that investigators must be selective about the regions of clothing they test based on what information is needed.
This study gives technicians greater insight into what collection methods are best for capturing touch DNA of either the “wearer” or “handler” of clothing. The researchers also claim that these results will allow investigators to determine trends in how much handlers and wearers donate to a DNA profile. Ideally, scientists could eliminate extraneous DNA that may arise from innocent people that encounter the handler or the wearer throughout the day. This is especially important, seeing that only 2 seconds of contact is required to be able to obtain a quality DNA profile from clothing. This knowledge can hopefully prevent false incrimination of people based on the sensitive touch DNA evidence.
Citation:
| Title | Touch DNA: impact of handling time on touch deposit and evaluation of different recovery techniques: An experimental study |
| Author(s) | Francesco Sessam Monica Salerno, Giuseppe Bertozzi, Giovanni Messina, Pietrantonio Ricci, Caterina Ledda, Venerando Rapisarda, Santina Cantatore, Emanuela Turillazzi, and Cristofor Pomara |
| Organization(s) | University of Foggia, Universitá degli Studi Magna Graecia di Catanzaro, University of Catania |
| Year | 2019 |
| Journal | Scientific Reports |
| Link | https://www.nature.com/articles/s41598-019-46051-9 |
