Scientists from the National Center for Forensic Science and Indiana University document soil degradation of ignitable liquids, potentially allowing analysts to overcome this issue with real evidence.
Featured image: “A prescribed fire on the North Rim of Grand Canyon National Park. NPS file photo.” from the National Park Service.
As many of us have learned from a young age, fire can be very dangerous. Just earlier this year, we got to see the horrific destruction caused by this element on the news during the Australian wildfires. Harnessing its devastating power, some of the most prolific criminals have used fire to kill many and cause millions of dollars of damage in crimes called arson.
Arson involves willfully and maliciously burning or attempting to burn an object or property. This is usually characterized by the use of an ignitable liquid to escalate the heat and spread of the fire to inflict more damage. There are many types of ignitable liquids that are classified into eight groups based on their composition of organic compounds (think, gasoline vs lighter fluid). These substances can be found in trace amounts, as most are usually consumed in the fire. Still, fire debris analysts can use separation techniques break a liquid down into its components and use these components to identify the class of an unknown ignitable liquid reliably. However, not all crime scenes are made equal, and some aspects of the scene can hinder analysis.

One major impact on ignitable liquids comes from the least expected of places: dirt. Porous surfaces like dirt can hold liquids easily, so it is natural to assume that dirt would be a good place to obtain ignitable liquids from. However, dirt also contains microbes that were discovered to degrade organic compounds. Many scientists have researched this microbial degradation and how it can impact some types of ignitable liquids. Dr. Dee Turner and her colleagues in Indianapolis and Florida wanted to go further. They wanted to generate a comprehensive database that logged degradation of several different ignitable liquids of all the major classes, listed out in Table 1. According Turner and her team, this would potentially prevent the misidentification of ignitable liquid evidence affected by these microbes.
| Class | Example (Used in experiment) |
|---|---|
| Gasoline | Chevron Unleaded |
| Oxygenated | E-Z Brush Cleaner |
| Petroleum Distillate (Heavy, Medium, Light) | Carburetor Cleaner (heavy), Paint Thinner (medium), Lighter Fluid (light) |
| Aromatic | Ortho Insect Spray Concentrated |
| Naphthenic-Paraffinic | Multipurpose Insect Cleaner |
| Isoparaffinic | ShellSol Mineral Spirits |
| Normal Alkane | Aura Lamp Oil |
| Miscellaneous | Chevron Aviation Gasoline |
Table 1. Major forms of ignitable liquids tested in this research.
Turner and her colleagues also tested the effect of weathering, or the gradual evaporation of the components in ignitable liquids. This phenomenon affects components with lower boiling points first and can cause all components to evaporate as time goes on. To make the weathered samples, the researchers filled a vial with 10 mL of ignitable liquids and gently heated them until the volume had decreased by 25%, 50%, 75%, 90%, and 95%. A small amount of the liquid after each heating session was removed and dissolved in carbon disulfide for testing. The degraded samples were prepared by spiking potting soil. The samples were placed in cans and stored for 0, 7, 14, and 21 days. The researchers then reopened the cans and hung an activated charcoal strip above the sample. The can was then resealed, and heat in an oven to evaporate the ignitable liquids and collect them on the strip. The strip was then placed into a vial with carbon disulfide for testing.
To test the samples, the researchers used a gas chromatography- mass spectrometry (GC-MS), a common methodology in fire debris units. Gas chromatography was used to separate the components in the liquids by their boiling points and chemical structure. Mass spectrometry acted as a detector, breaking the components into their unique ions and measuring the ratio of their sizes to their charges.
The researchers found that, across all ignitable liquids, weathering resulted in the loss of the components based on their boiling points, which confirms previous research. The components with the lowest boiling points vanished first, and as heating continued, components with higher boiling points began to fade. These changes did not significantly impair the scientists’ abilities to identify the important components in the ignitable liquids.
As for the degraded samples, the loss of components was based on chemical structure, regardless of boiling points. The bacteria tended to attack any present n-alkanes first, followed by less-substituted alkylbenzenes in order of increasing functional groups. The bacteria affected all of the ignitable liquids in this way.
However, the degradation impacted some classes of liquids more than others. Gasolines, petroleum distillates, and the oxygenates were among the most affected. For an ignitable liquid to be classified as gasoline, its chromatogram must show presence of a particular type of alkylbenzene (C3-alkylbenzene), but degraded samples lost this component. As such, a degraded gasoline sample would end up being misidentified as another ignitable liquid, or even dismissed! This also occurred with high-petroleum distillates. The loss of their characteristic n-alkanes made their chromatograms appear to be those of the paraffinic groups.
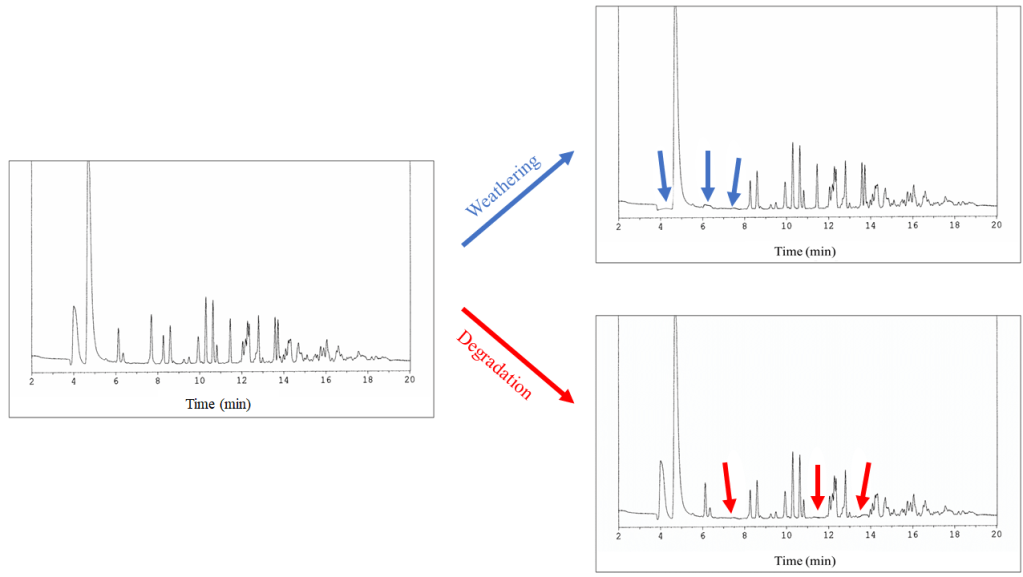
Dee and her team used their data to update the existing databank of ignitable liquids, known as the Ignitable Liquids Reference Collection (ILRC), created by the National Center for Forensic Science. Adding the data of ignitable liquids subjected to weathering and soil degradation will hopefully allow forensic scientists to properly identify impacted evidence. As discussed before, degradation can distort the chromatograms of ignitable liquids, making them hard to identify and may even cause them to be misclassified. While fire debris evidence such as ignitable liquids is only circumstantial, it can still be powerful in court. A misidentification could potentially cause false incrimination or the release of a guilty person. Making sure analysts understand how different environmental circumstances affect their samples will allow them to take these conditions into account and correctly analyze evidence.
| Title | A Comprehensive Study of the Alteration of Ignitable Liquids by Weathering and Microbial Degradation |
| Author(s) | Dee A. Turner, Mary Willaims, Michael A. Sigman, John V. Goodpaster |
| Organization(s) | Indiana University Purdue University Indianapolis, National Center for Forensic Science |
| Year | 2018 |
| Journal | Journal of Forensic Sciences |
| Link | https://onlinelibrary.wiley.com/doi/abs/10.1111/1556-4029.13527 |

One thought on “How does dirt affect ignitable liquids?”